 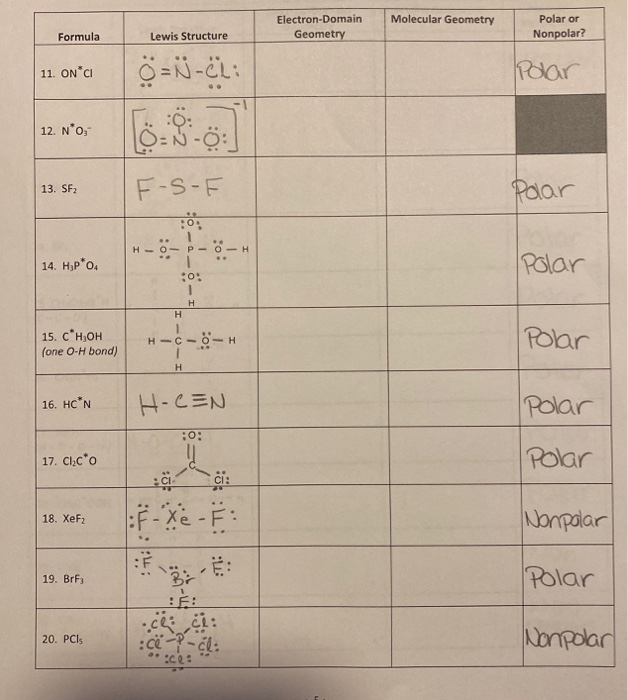
In the Lewis Structure of SF2, the central atom forms two bonds with two Fluorine atoms and has two lone pairs of electrons. 
The bridging F atom is coplanar with the cation with a bridge angle of 140.8 (4)°. The structure consists of XeF3+SbF6- units with a close contact of 2.485 (10) Å between the Xe atom of the T-shaped cation and an F atom of the octahedral anion. 
The shape of H3O+ is pyramidal as the 3 hydrogen atoms lie at three corners of oxygen as a triangle, and one lone pair of electron on oxygen attains pyramidal shape to H3O+. The three atoms bond at 90° angles on one side of the central atom, producing the T shape. How many lone pairs does ibr3 have? In an AX3E2 molecule, the two lone pairs occupy two equatorial positions, and the three ligand atoms occupy the two axial positions as well as one equatorial position.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
